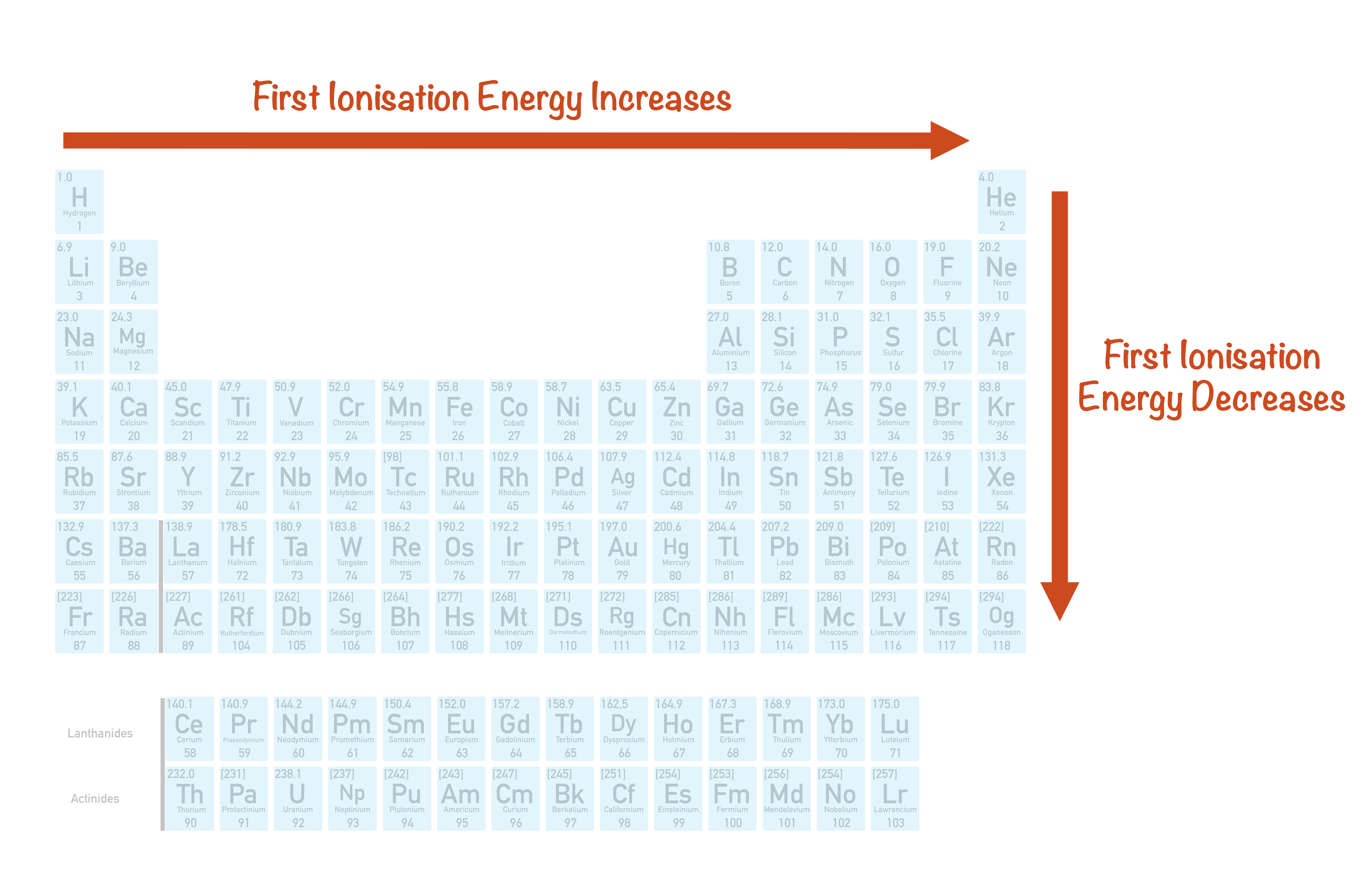
In other words, even though an element group might break a trend, the elements within the group display periodic properties. However, the behavior of the noble gases is periodic. The periodic table, also known as the periodic table of the elements, arranges the chemical elements into rows ('periods') and columns ('groups'). The noble gases are an exception to the trend since these elements have filled electron valence shells and electron affinity values approaching zero. Nonmetals usually have higher electron affinities than metals. The inorganic chemistry p block elements properties like that of other block elements are greatly influenced by their ionization enthalpy, atomic size, electronegativity, and electron gain enthalpy. Electron affinity increases moving across a period and decreases moving down a group. The elements that are placed in group-13 to group-18 of the periodic table compose the p-block. Electron Affinity - This is a measure of readily an atom accepts an electron.Atom and ion sizes shrink moving across a period because the increasing positive charge of the nucleus pulls in the electron shell. Although it might seem like increasing the number of protons and electrons in an atom would always increase its size, the atom size doesn't increase until a new electron shell is added. Ionic radius is the distance for ions of the atoms and follows the same trend. Atomic radius decreases moving left to right across a period and increases moving down a group. Modern Periodic Table Questions And Answers - Practice questions, MCQs, PYQs, NCERT Questions, Question Bank, Class 11 and Class 12 Questions, NCERT Exemplar Questions, and PDF Questions with answers, solutions, explanations, NCERT reference, and difficulty level in Modern Periodic Table chemistry. They are organized in a tabular format, where a row represents a period, and a column represents a group. 
Atomic Radius - This is half the distance between the middle of two atoms just touching each other. The periodic table is an arrangement of all known elements in order of increasing atomic number and recurring chemical properties.Electronegativity - A measure of how readily an atom forms a chemical bond. Electronegativity increases moving left to right across a period and decrease moving down a group.Ionization energy increases moving left to right across the table and decreases moving down a group. They both arranged the elements by their mass and proposed that certain properties periodically reoccur. Mendeleev created the first periodic table and was shortly followed by Meyer. Ionization Energy - This is the energy needed to completely remove an electron from an atom or ion. The periodic law was developed independently by Dmitri Mendeleev and Lothar Meyer in 1869.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
